Are Food Labels Lying to You? The Real Science Behind Calorie Counts
Ever wondered how scientists know that a slice of bread contains exactly 79 calories, or that a tablespoon of olive oil clocks in at 119? It seems almost impossible to pinpoint so correctly. for something as messy and unpredictable as food. It feels like someone, somewhere, must have literally sat down and counted every single one. But the truth is way more fascinating, and a lot more scientific, than that.
Calorie counts on nutrition labels aren’t guesses or rough estimates. They’re the result of real, carefully conducted science — a blend of chemistry, physics, and nutrition research that has been refined over more than a century. And understanding where those numbers actually come from? It genuinely changes the way you read a label. So let’s pull back the curtain on what’s really happening from the lab bench to your kitchen table. In this post, we’ll break down the core methods used to determine calorie counts and why the result matters for everyday cooking and budgeting meals.
https://www.fda.gov/food/nutrition-facts-label/calories-nutrition-facts-label

First Things First: What Even Is a Calorie?
Before we get into the how, let’s nail down the what.
A calorie is simply a unit of energy. Technically, what you see on a food label is actually a kilocalorie (kcal) — but the two words are used so interchangeably that most people (and most labels) just say “calories.” One kilocalorie is the amount of energy needed to raise the temperature of one kilogram of water by one degree Celsius.
Here’s a fun little translation:
- 250 Calories on a label = 250 kilocalories = 250,000 small calories
Your body doesn’t “feel” that in any direct way — but it gives scientists a universal, measurable way to compare the energy in different foods. When you eat, your body breaks down the chemical bonds in food and releases the energy stored inside them. That energy is what keeps your heart beating, your muscles moving, and your brain running.
The challenge? You can’t see or touch energy directly. You can only observe what it does — like raising the temperature of water. And that simple observation is the entire foundation of calorie science.
Bomb Calorimetry vs. The Atwater System: Which Is More Accurate?
There are two main approaches to figuring out how much energy is hiding in your food:
1. Bomb Calorimetry (the direct method) This is the original, gold-standard technique. A small, dried sample of food is placed inside a sealed metal chamber called a “bomb,” filled with oxygen, and ignited. The heat released from combustion raises the temperature of surrounding water, and sensors measure exactly how much. Precise, dramatic, and genuinely cool — more on this below.
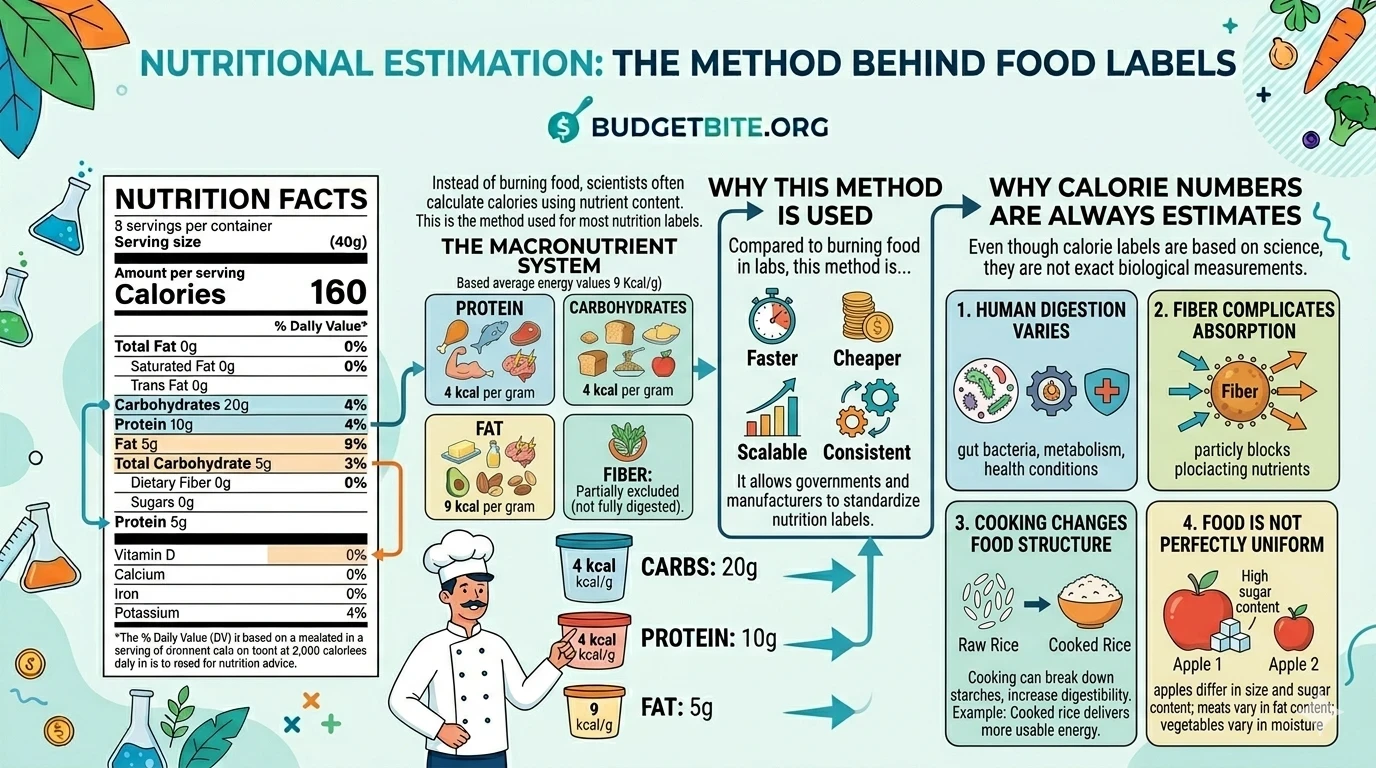
2. Nutritional Estimation (the indirect method/Atwater method) For everyday food labeling, scientists analyze the macronutrient makeup of a food (how much carbohydrate, fat, and protein it contains) and apply standard energy conversion factors to estimate the calorie count. This is the more practical approach for labeling millions of products efficiently.
Both methods have their strengths. Both have trade-offs. And understanding them helps explain why calorie counts are best thought of as reliable estimates -not absolute biological facts. Your body, after all, is not a sealed metal chamber.

The Spark of Science: What is a Bomb Calorimeter?
Here’s where it gets fun.
Scientists take a small, carefully dried sample of food — say, a little bit of freeze-dried chicken breast, or a compressed crumb of bread — and seal it inside a thick-walled metal chamber called a bomb calorimeter. The chamber is flooded with pure oxygen under high pressure, and the sample is ignited with an electric spark.
What happens next? The food combusts completely — not a slow burn, but a rapid, total release of all its stored chemical energy. The heat from that explosion transfers to the water surrounding the chamber, and sensors track exactly how much the temperature rises. From that rise, scientists calculate the food’s energy content with impressive accuracy — typically within 0.1 to 0.2%.
In just a few seconds, a pellet of food releases all the energy it took a living organism to build. And that release gets captured, measured, and eventually translated into the number printed on your label.
The science behind it comes straight from the First Law of Thermodynamics: energy can’t be created or destroyed — only transformed. When food burns, the chemical energy stored in its molecular bonds — the bonds holding together fats, proteins, carbohydrates — converts entirely into heat. That heat is measurable, and that measurement tells scientists exactly how much energy was packed inside.

Step-by-Step: How a Calorimeter Calculates Food Energy
Each bomb calorimetry test follows a precise, standardized routine designed to keep variables as controlled as possible:
Step 1 — Sample Preparation The food is freeze-dried to remove all moisture. (Water doesn’t burn, and its presence would throw off the measurement entirely.) Once dry, the sample is ground into a fine powder and compressed into a small pellet — usually between 0.5 and 1.5 grams.
Step 2 — Loading and Sealing The pellet goes inside the bomb — a sealed stainless steel vessel built to withstand extreme pressure and heat. A thin ignition wire is threaded through the pellet to carry the electric current that starts the combustion.
Step 3 — Pressurization All air is purged from the chamber and replaced with pure oxygen at around 3.0 MPa — about 30 times normal atmospheric pressure. The high-oxygen environment ensures the sample burns completely, leaving nothing behind to skew the results.
Step 4 — Equilibration The sealed bomb is submerged in a precisely measured volume of water, gently stirred for about five minutes, and allowed to stabilize at a baseline temperature. That starting temperature is critical for the final calculation.
Step 5 — Ignition An electric current passes through the wire and — whoosh — combustion happens in seconds.
Step 6 — Measurement Sensors record the water temperature continuously for 10 to 15 minutes, tracking the rise until it levels off. The difference between the starting and ending temperature — combined with the known heat capacity of the calorimeter — gives scientists the exact energy released.
The result is called the gross energy of the food: the total chemical energy from complete combustion. But this isn’t quite the same as the number you see on a nutrition label. Food scientists take one more step, adjusting gross energy for digestibility and the efficiency of nutrient absorption, to arrive at metabolizable energy — the calories your body can actually use, the number that ends up on your label.
Not All Calorimeters Are Built the Same
While the core principle is always the same — ignite a sample, measure the heat — laboratories use several different calorimeter designs depending on how much precision they need and what environment they’re working in.
Isoperibol calorimetry is the most common setup for commercial food testing. The outer jacket stays at a constant temperature while the inner water bath rises naturally during combustion. A small mathematical correction accounts for any heat exchange between them. It’s accurate, practical, and well-suited for routine testing.
Adiabatic calorimetry takes precision further. The outer jacket continuously adjusts to match the inner bath temperature exactly, creating a fully isolated system. It’s preferred for high-stakes research where even tiny variations matter.
Isothermal calorimetry keeps the entire system at a constant temperature and instead measures the energy needed to maintain that steady state during combustion. This works especially well for slow chemical reactions or biological processes.
Dry static jacket calorimetry ditches the water bath entirely, sitting the bomb inside a solid aluminum block with built-in temperature sensors. Faster to set up, better for high-throughput industrial settings — with a slight trade-off in precision that most practical applications can live with.
The Indirect Method: Where Does the “4 Calories Per Gram” Rule Come From?
Running a full bomb calorimetry test on every food product would be time-consuming and expensive at scale. For most everyday nutrition labeling, scientists instead estimate calorie content by analyzing the macronutrient breakdown and applying standard energy conversion factors.
Those factors — developed over decades of research — look like this:
- Carbohydrates: ~4 kcal per gram
- Protein: ~4 kcal per gram
- Fat: ~9 kcal per gram
- Alcohol (when present): ~7 kcal per gram
So if a food contains 20g of carbs, 5g of protein, and 8g of fat per serving, the estimated calorie count would be: (20×4) + (5×4) + (8×9) = 172 kcal.
This system is called the Atwater factors, named after 19th-century chemist Wilbur Atwater who developed them. It’s efficient, consistent, and forms the backbone of virtually all modern nutrition labeling in the U.S. and many other countries.
One nuance worth knowing: dietary fiber is technically a carbohydrate, but because humans can’t fully digest it, it contributes far less usable energy than other carbs. More sophisticated versions of the Atwater system assign fiber a lower energy value — which is why “net carbs” calculations subtract fiber from total carbs.
The Moisture Factor (This One Surprises People)
Here’s a detail that often flies under the radar: water has no calories, but it makes up a huge proportion of many foods by weight.
A raw chicken breast is roughly 75% water. Cook it, and much of that moisture evaporates — meaning the same portion weighs less but still delivers the same calories. The result? Cooked chicken appears more calorie-dense per gram than raw chicken, even though you’re getting the same total energy.
This is why bomb calorimetry always starts with freeze-dried samples. Removing moisture first ensures the measurement only captures the energy-containing components. Ignore moisture differences when translating lab results to real-world labels, and your estimates can end up meaningfully off from what people are actually eating.
So… How Accurate Is the Number on the Package?
Here’s something important to keep in mind: the calorie count on your label is a reliable, standardized estimate – not not an always perfect 100% biological readout of what your body will extract from every bite. It’s a reliable, standardized estimate – and that distinction matters.
Human digestion is variable. The same food can yield slightly different amounts of usable energy depending on your gut microbiome, how thoroughly you chew, your individual digestive efficiency, and even how the food was cooked. Cooked starches, for instance, are more digestible than raw ones — which is part of why humans extract more calories from cooked food than the same food eaten raw.
None of this means calorie labels are unreliable or not worth paying attention to. It means they’re a well-constructed tool for comparison and planning — not a precise personal metabolism tracker. Used correctly, they’re genuinely useful. Treated as an exact science, they can give a false sense of precision that doesn’t quite match reality.

How to Actually Use Calorie Info Well
Understanding the science makes you a smarter label reader. A few practical principles worth keeping in your back pocket:
Always check the serving size first. The calorie number is meaningless without knowing the portion it applies to. Many packages contain two or three servings — which means those listed calories might be for a fraction of what you’d actually eat in one sitting.
Look at the full macronutrient picture. Calories tell you about energy density, but they don’t tell you about satiety, nutrient quality, or how a food affects your blood sugar. A 200-calorie serving of nuts hits your body very differently than a 200-calorie serving of sugary cereal.
Factor in cooking and preparation. If you’re tracking carefully, remember that boiling, roasting, frying, and steaming can all affect a food’s energy availability and nutrient content in different ways.
Treat labels as a guide, not a rulebook. Your energy needs are shaped by your age, body composition, activity level, and health status. A number on a package can inform your choices — it can’t make them for you, and it shouldn’t override your own hunger and fullness cues.
Think about nutrient density alongside calorie density. A food that’s lower in calories but packed with vitamins, minerals, fiber, and protein is almost always a smarter choice than something that’s low in calories but nutritionally hollow. The calorie count is just one piece of a much bigger puzzle.

The Takeaway
The next time you glance at a nutrition label, there’s a whole chain of careful science behind that number. Somewhere along the line, a version of that food was freeze-dried, compressed into a tiny pellet, sealed inside a pressurized metal chamber, and literally set on fire — all so researchers could measure, with remarkable precision, exactly how much energy it contained.
That number was then adjusted, standardized, and converted into the calorie count printed in bold on your package. It accounts for digestibility and macronutrient composition, and it’s designed to be a consistent, comparable reference point across millions of different products.
It’s not magic. It’s not a guess. It’s chemistry – translated, one combustion test at a time, into something genuinely useful for the way you eat every day.
https://www.who.int/news-room/fact-sheets/detail/healthy-diet
https://www.scientificamerican.com/article/how-do-food-manufacturers/

This was such a fascinating and well-explained deep dive into something most of us take for granted. I love how you break down the science in a way that feels approachable without oversimplifying it. The explanation of bomb calorimetry was especially engaging, it’s wild to think our food is literally analyzed through controlled combustion! You also did a great job clarifying the difference between gross energy and what our bodies actually use, which adds important context to calorie labels. The section on moisture and cooking effects was a real eye opener too. Overall, this post strikes a great balance between scientific detail and practical takeaways. It definitely changes how I’ll look at nutrition labels moving forward, more as useful guides than exact numbers. Really insightful and thought provoking read.
Thanks Andrejs, I had always been skeptical of the numbers on those labels, thought it was impossible to pinpoint so accurately of just plain made up. I find it fascinating how much there is behind it, and how accurate they actually get.